Hesitation and hope: The vaccine
Pan American Health Organization
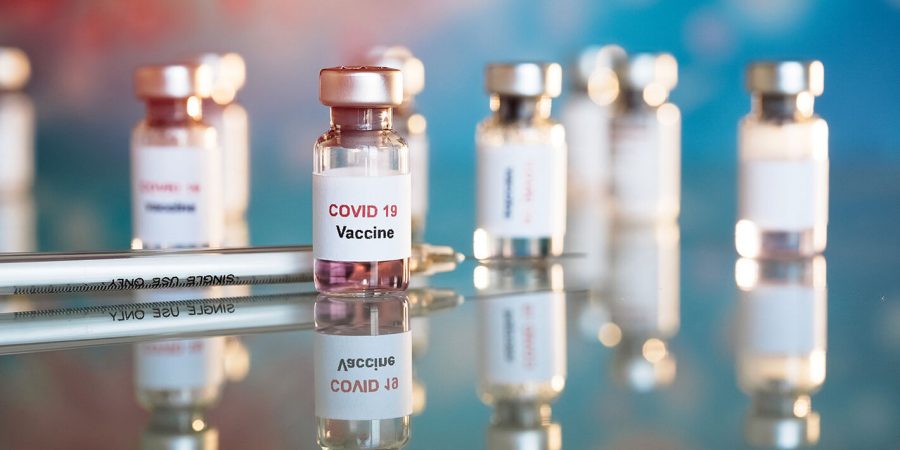
Majority of COVID-19 vaccines currently getting developed will be given in two doses a little under a month apart.
December 4, 2020
The past 10 months have felt like a lifetime and we are all longing for a sense of normalcy. The recent COVID-19 vaccines are giving us hope.
According to The New York Times, there are 78 vaccines currently undergoing clinical trials. Seven of these are approved for limited use. And many countries are beginning to distribute vaccines from various providers for emergency use.
After proving to be at least 90% effective when fighting COVID-19, the vaccines we may start to see roll out in the US include Pfizer, BioNTech, and Moderna. If approved by the Food and Drug Administration (FDA), many of these will be given starting mid-December and as we approach the new year.
In the U.S. there is no federal distribution plan, instead decisions are handed over to the governors. Each state will get an amount of vaccines and will take their own course of action.
For Colorado, this means three phases.
| Phase 1 |
|
| Phase 2 |
|
| Phase 3 |
|
Source: Coloradoan(modified)
Stephan Sears, a gastroenterologist is included in the first release of Colorado’s vaccine plan. As a healthcare worker who sees dozens of COVID-19 patients he has been fearful of bringing the virus home to his family.
“I’m very excited about it [the vaccines] because I work in an environment where there’s a lot of exposure to COVID-19,” says Sears.
Around three fourths of the population would need to be vaccinated to approach a more normal world. A concern Sears has pertaining to the vaccines is people not receiving both doses of the vaccine.
He explains, “the problem is that it’s two shots, and you feel pretty sick, like a day after it. So I think a lot of people who will get the vaccine, the first shot, they might not come back for their second vaccine.”
Another worry Sears voices is the unknown longevity of the vaccine. With the accelerated development of the vaccines, we are unsure how long the antibodies will last. Months or years down the road, we may find certain vaccines prove to be more reliable, narrowing down the options.
The injection of the vaccine helps to stimulate the growth of white blood cell antibodies. With the vaccine, our bodies will be able to defend against the virus.
“Our body will recognize those as foreign, so then it will make white blood cells that have the capacity and antibodies to destroy those spike proteins so if you get the virus it will recognize those spike proteins and then the virus can’t invade you,” Sears simplifies.
With many different vaccine companies, we still wonder which one to choose. The reality is, hospitals and medical providers will be given a supply of vaccines regardless of who manufactured them.
There can be lots of hesitation surrounding the quickly generated vaccines which is completely understandable. The truth is, if the vaccines continue to reduce risk and are confirmed to be safe through more trials, we have hope to slowly return to a pre-pandemic world.